PBM Light
Photobiomodulation (PBM Therapy) utilizes red light and near infrared radiation over injuries or lesions to stimulate healing and relive pain without sensation or side effects.
Sun light is a mixture of seven different colors, whereas Laser Light is always of one single color. (Monochromatic). Sunlight is disorderly whereas Laser Light is very orderly (Coherent). Sunlight is not parallel whereas Laser Light runs parallel (Collimated). The healing properties of Laser Light comes from these differences.
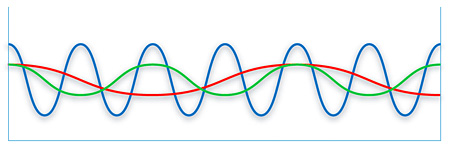
Sunlight - many different colours

LED Light - one colour (monochromatic) and waves not in phase (non-coherent)

Laser - one colour (monochromatic) and waves in phase (coherent)
 Featured Testimonials
Featured Testimonials




